
Note that pH is unit-less and there is a space before the degree symbol. The ever present solvent in cells is water physiological conditions assume a pH of 7.4 and temperature of approximately 37 ☌. The two main groups are the hydrophobic amino acids(water fearing) and the hydrophilic amino acids(water loving). The 20 common amino acids can be classified by their side chains. The a-carbons on either side are locked into specific geometries that will later form a-helices and b-sheets in proteins. The alignment of the p-orbitals between the amide nitrogen and the carbonyl carbon and oxygen facilitates the sharing of electrons, strengthening the bonds, making them rigid pseudo-double bonds. When AAs are joined, the resulting peptide bond has a planar geometry (Figure 4). The overlapping p-orbitals stabilize a planar geometry. Errors in synthesis ( mutations) are reported like this: Ser2 ➔ Thr this means the serine in position 2 was mutated to a threonine. In the cell, proteins are synthesized from the amine terminus to the carboxy terminus, and we maintain this convention in reporting sequences. The sequence above (in 3-letter code) is Ala1-Ser2-Gl圓-Leu4. Peptides and proteins are written and numbered left to right, starting at the N-terminus and ending at the C-terminus. The –NH 2 group of the first peptide is called the amine or N-terminus the –COOH group of the last peptide is the carboxy or C-terminus. In Figure 3, four amino acids–alanine, serine, glycine and leucine–are connected by peptide bonds (blue). Amino acids joined by peptide bonds in blue. In literature, a particular AA may be referred to as a residue e.g., “The lysine residues in histones are acetylated to promote dsDNA packaging.” Figure 3. Short sequences less than 50 AAs are called peptides and sequences greater than 50 AAs are referred to as proteins. 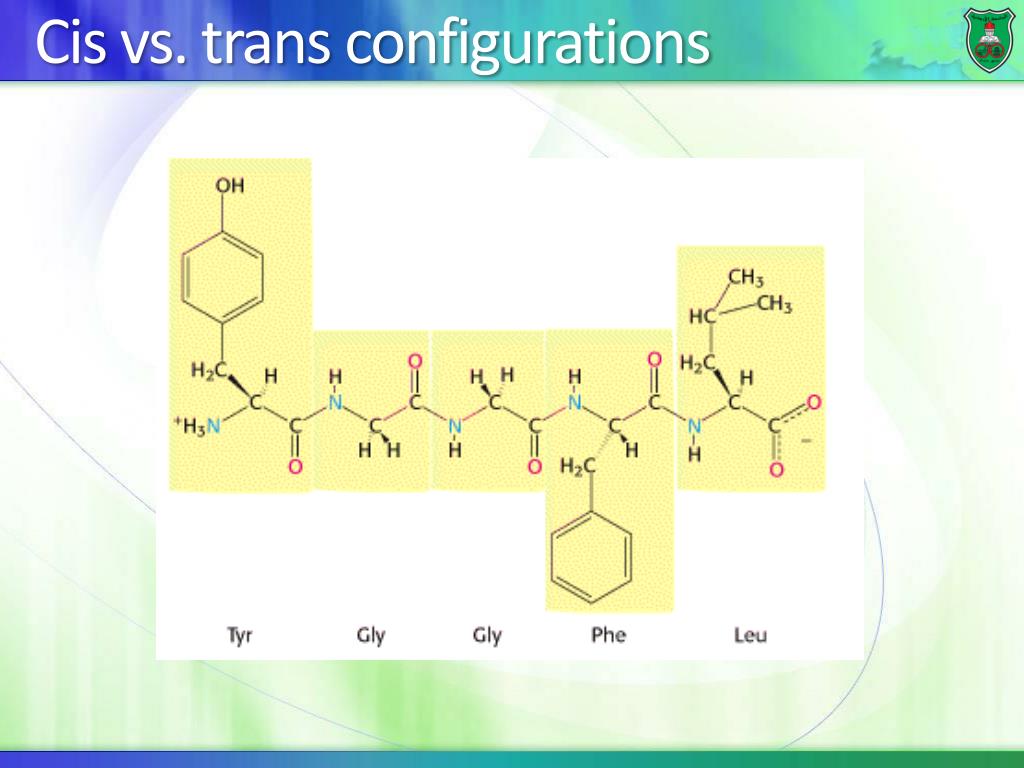
The newly formed amide bond or peptide bond is formed by a dehydration reaction. Image Source: Wikimedia CommonsĪmino acids are the monomeric building blocks of proteins the cell builds proteins by joining amino acids to each other from carboxylic group to amine group. Reversing or inverting the structure will have implications on the wedge or hashed ( dashed) marked bonds (Figure 3). Ask the instructor to provide the stereochemistry whenever possible. Course instructors will draw structures many different ways. If you are confused about stereochemistry, refresh yourself on Fischer projections from organic chemistry. Biochemists focus on L-amino acids, as depicted in Figure 1 (wedge bonds), but D-amino acids are relevant for pharmaceutical applications. The R group, or side chain, is unique for each amino acid as a result, the a-carbon is a chiral center, except for glycine where R = hydrogen. Also, some amino acids have unique traits that add to their functionality Carbon SkeletonĪll of the amino acids are composed of an amine(-NH 2) and a carboxylicacid (-COOH) linked by a central a-carbon (Figure 2). To learn pH effects or the physicochemical properties of individual AAs, it’s best to try to understand the process and how changing the microenvironment will alter how an AA will behave. Also be keen on the nomenclature and conventions presented to you in class. Keep the table with you like the periodic table of elements in chemistry, you will refer to this AA table throughout the course. The three letters in parentheses are the 3-letter shorthand, and the letter in the red circle is the 1-letter shorthand: e.g., arginine, Arg, R. To read Figure 1, the AA in the top left is arginine. Note that selenocysteine (Sec, U) is not a common amino acid and can be skipped. A table of the common amino acids is provided in Figure 1. Use flash cards, whiteboards, or any other method of repetitive memorization. To learn the structures, names, and shorthand, the best method here is memorization. That is a daunting task for 20 amino acids. Most biochemistry courses will require you to know the following: the amino acid name, the structure, the pKa of ionizable hydrogens, and both the 3-letter and 1-letter shorthand. Always keep in mind, structure gives function. 
Errors in amino acid placement do occur and can lead to cell death in some instances. The AAs are strung together by ribosomes that read the instructions given to it by the mRNA. These proteins are made up of smaller building blocks called amino acids (AA). Proteins are complex biomolecules that perform critical roles in the cell. When genes are expressed, the DNA is transcribed into mRNA that is then translated into proteins. In biochemistry, DNA holds the genetic code for life.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
